UCL researchers collaborate with an international research team to improve the efficiency of low-cost, sustainable batteries which could power the future. By Melike Temizturk
Have you ever felt nostalgic about the technology that defined your childhood? Perhaps you miss wrapping your finger in the coiled cable of a landline, rolling the tape back into a cassette, or clicking the buttons on a chunky mobile phone. We grew up with these devices, but the world seems to have grown out of them, almost overnight. What facilitated the rapid shift towards the lighter, wireless electronics we know and use today?
It's a natural consequence of innovations in electrochemical storage solutions, in particular, the lithium-ion battery (LIB). Since their commercialisation in 1991, they have cornered the market on portable electronics; chances are, whatever device you’re currently reading this article on is powered using a LIB. Its huge energy output is the result of the electrochemical reactions the small, charged lithium-ions induce as they move from the cathode, into the anode, and back again. However, the meteoric rise to the top of the massive battery market comes at a cost. Some have raised sustainability concerns about using lithium, which is highly reactive and inefficiently reclaimed from used parts. Furthermore, the exploitation of labourers, including children, to mine the cobalt needed for the cathode is a high price to pay for consumer electronics.
A recent collaboration between UCL, the Agency for Science, Technology and Research in Singapore and the Harbin Institute of Technology in China has brought us a step closer to realising low-cost, sustainable energy storage. Researchers have synthesised a highly electrocatalytically active cathode as an alternative to the precious metals electrocatalysts currently used in zinc-air batteries (ZABs). Oxygen binds better to the surface of the electrocatalyst, improving the rate of the redox reactions that power the battery [1]. Researchers hope to one day attain the huge theoretical energy density of metal-air batteries, which surpasses even that of LIBs.
ZABs have effectively outsourced the electrochemically active oxygen by allowing it to diffuse in through the porous cathode. Here, it is reduced as part of the discharging action. This oxygen reduction reaction (ORR) is notoriously slow as it is not very thermodynamically favourable. Consider the strong O=O double bond in oxygen, which requires huge amounts of energy to overcome. ZABs accomplish this by activating the cathode using an electrocatalyst, which lowers the activation energy (the energy barrier preventing the breaking of the bond) allowing the ORR to proceed faster.
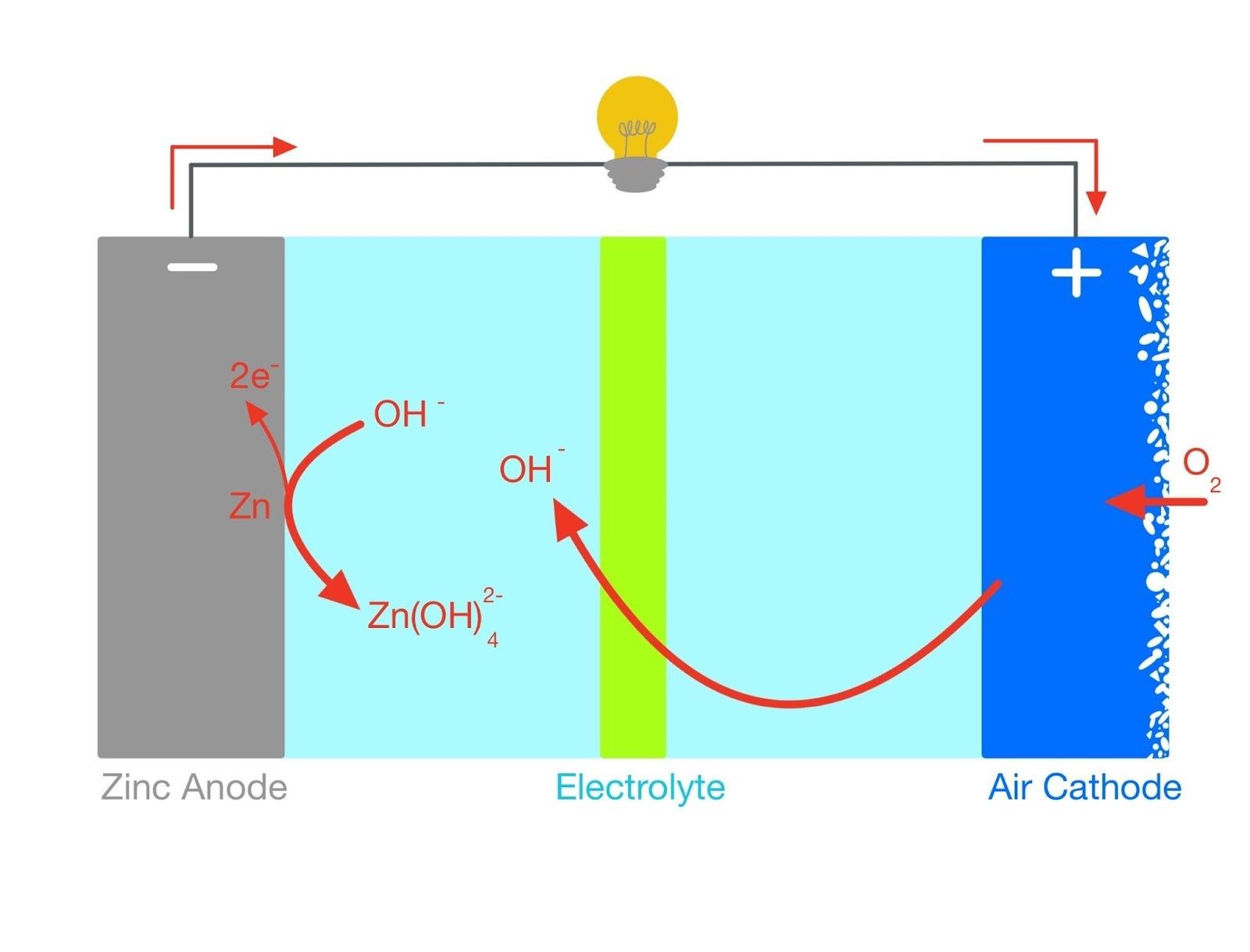
To synthesise the electrocatalyst, researchers employed graphitic carbon nitride (C3N4) as a scaffold for the polymerisation of dopamine (yes, the happy hormone). The alternating layers of C3N4 and polydopamine served to increase the surface area for oxygen adsorption/desorption. Cobalt ions were introduced in situ to promote the growth of metal-organic frameworks (MOFs). When heated at 900°C, these MOFs were reduced to cobalt nanoparticles and carbon nanosheets which increased electrical conductivity throughout the structure. This not only improves the ORR when the battery is discharging but also catalyses the reverse reaction, the oxygen evolution reaction (OER) during charging.
This bifunctionality sets this electrocatalyst apart from those that are currently available for use in zinc-air batteries. The two most common are platinum/carbon and iridium/carbon which catalyse the ORR and OER respectively. The cobalt/carbon electrocatalyst exhibited excellent activity and outperformed both, with minimal performance loss after cycling for 600 hours.
Electrocatalysis could be the key to unlocking the full energy capabilities of zinc-air batteries, especially with recent efforts to improve the anode. Still, many have doubted the feasibility of scaling up a technology that would require constant airflow to function. The Japanese electronics manufacturer, Sharp Corporation, has adapted to this by implementing their ZABs in outdoor renewables, like solar panels and wind turbines. This ingenious idea could cement the role of ZABs in the future of sustainable energy production and storage.
References:
[1] Ferraz, B., Kong, J., Li, B., Neng Tham, N., Blackman, C. and Liu, Z., 2022. Co/N nanoparticles supported on a C3N4/polydopamine framework as a bifunctional electrocatalyst for rechargeable zinc-air batteries. Journal of Electroanalytical Chemistry, 921, p.116702.